

✓ Good Distribution Practice for Medical Device (GDPMD) ✓ Establishment License, Medical Device Authority (MDA) ✓ Laboratory Accreditation
(10 IN 1) COVID-19/HMPV/RSV/ADV/ FLU A&B/HPIV1+3/HPIV2/MP/RHV ANTIGEN COMBO RAPID TEST KIT
(MDA Registration No: IVDC3608225-210153)
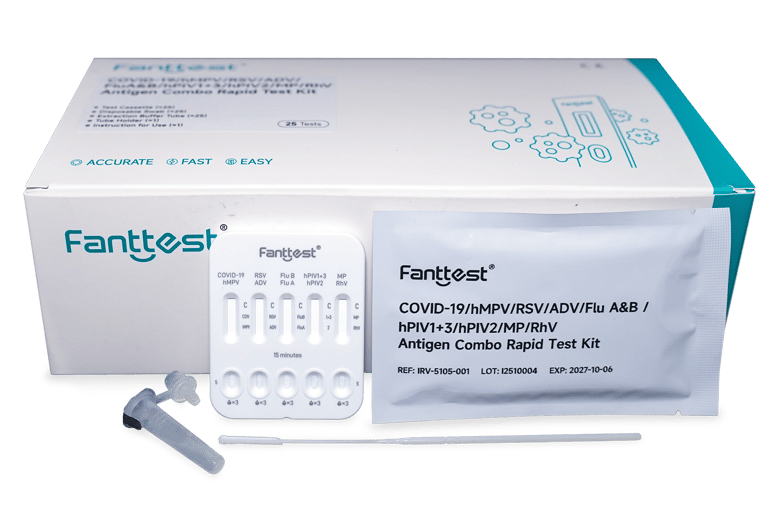

COVID-19/hMPV/RSV/ADV/Flu A&B/hPIV1+3/hPIV2/MP/RhV Antigen Combo Rapid Test Kit is a lateral flow immunoassay for the qualitative detection of COVID-19, respiratory syncytial, Adenovirus, M.pneumoniae, Rhinovirus, influenza A+B, Parainfluenza 1+3, Parainfluenza 2, human metapneumovirus antigens in nasopharyngeal swabs. The symptoms of respiratory viral infection due to COVID-19, respiratory syncytial, Adenovirus, M.pneumoniae, Rhinovirus, influenza A+B, Parainfluenza 1+3, Parainfluenza 2, human metapneumovirus can be similar. The test is intended as an aid in diagnosis of symptomatic individual meeting the case definition for COVID-19 in the first 7 days of symptom onset and meeting the case definition for respiratory syncytial, Adenovirus, M.pneumoniae, Rhinovirus, influenza A+B, Parainfluenza 1+3, Parainfluenza 2, human metapneumovirus in the first 4 days of symptom onset.


